Day 2 :
Keynote Forum
Lallan Giri
Biologic Resources LLC, USA
Keynote: Conjugate rPA-PGA anthrax vaccine induces protective antibodies
Time : 10:00-10:40

Biography:
Lallan Giri is currently the CEO of Biologics Resources LLC (BRLLC) which is a Vaccine and Biopharma Company focused on the development of biodefense vaccines for adults and children. He is a Vaccinologist and has made contributions to the development of several pediatric vaccines as Director at Glaxo Wellcome, Director at Sanofi Pasteur, Vice President at Emergent Bio-solutions, and CEO at BRLLC. The current ongoing research projects at BRLLC include work on “Biodefense vaccines and therapeutics”.
Abstract:
Anthrax is a lethal infectious disease caused by Bacillus anthracis. The two major virulence factors of B. anthracis are the poly-γ-D-glutamic acid (PGA) capsule and the exotoxin. Three components of the exotoxin form two bipartite toxins causing edema and organ failure. The anti-phagocytic PGA capsule disguises bacilli from macrophages, allowing unimpeded growth in the host. PA is a target of anthrax vaccine development as it elicits a toxin-specific protective immune response; however PA based anthrax vaccines only address toxin-induced disease and not capsule mediated virulence. Recent effort has been made to include PGA in 2nd generation anthrax vaccines, resulting in a promising vaccine providing comprehensive protection. Antibody to PGA fully neutralizes the capsule but PGA must be conjugated to a carrier protein to provide immunogenicity against both antigens. We have undertaken advanced development of an rPA-PGA conjugate vaccine for adult and pediatric populations. Our hypothesis is that by targeting the two virulence factors of B. anthracis, we can create an anthrax countermeasure that is more comprehensive and efficacious than currently available vaccines. We have shown by ELISA that our conjugate vaccine candidate produces significant levels of antibody to PA and PGA in mouse and rabbit immunogenicity studies. Compared to the approved anthrax vaccine, antibodies produced in response to the rPA-PGA vaccine are more protective in the toxin neutralization assay. We have produced cGMP vaccine and performed a challenge study in rabbits with this vaccine lot, demonstrating 100% protection against lethal aerosol B. anthracis spore challenge in vaccinated rabbits. There is also an increasing need to develop an anthrax vaccine for use in children. We have developed a neonatal rabbit model to simulate the pediatric immune response, showing three week old rabbits generate antibody to both antigens in response to immunization with rPA-PGA that is neutralizing in TNA.
Keynote Forum
Leondios G Kostrikis
University of Cyprus, Cyprus
Keynote: Development of vaccines using genetically engineered Inoviruses
Time : 10:40-11:20

Biography:
Leondios G Kostrikis is a Professor at University of Cyprus. He completed his BSc and PhD at New York University and his Post-doctoral training at Aaron Diamond AIDS Research Center of Rockefeller University. He joined the Rockefeller University as an Assistant Professor in 1999 and the University of Cyprus as Professor in 2003. He was a Fulbright, Elizabeth Glazer Pediatric AIDS Foundation and Aaron Diamond Foundation Scholar. He has directed over 20 competitive grants from the NIH and the European Commission. He is an Editorial Board Member of 12 international journals.
Abstract:
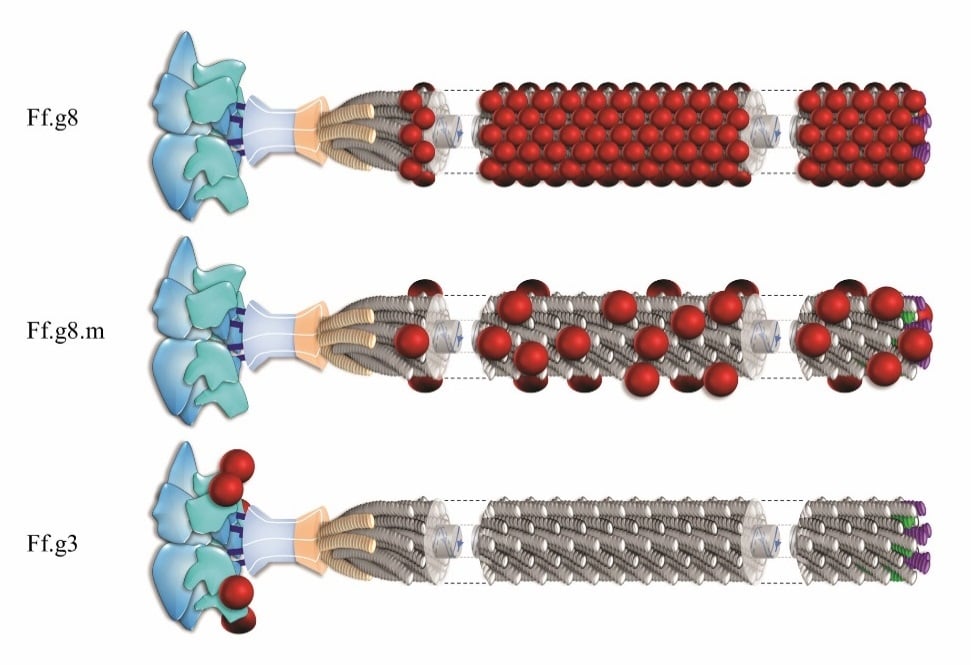
An effective vaccine will likely depend on success in designing immunogens that elicit potent neutralizing antibodies to naturally circulating strains of infectious agents including viruses. In the past, antigen-presenting viral vectors have been extensively used as vehicles for the presentation of antigens to the immune system in numerous vaccine strategies. In previously known vaccine development efforts, two main viral vectors have been used as antigen carriers: Live attenuated vectors and virus-like particles (VLPs); the former, although tends to be highly effective in animal studies, raises safety concerns and the latter is less effective to induce neutralizing antibodies. For more than two decades, Inoviruses have also been utilized as antigen carriers in several vaccine studies. Inoviral vectors are important antigen-carriers in vaccine development due to their ability to present an antigen on their outer architecture in many copies and to their natural high immunogenicity. Numerous fundamental studies have been conducted, which have established the unique properties of antigen-displayed inoviral vectors. The recent isolation of new, potent neutralizing monoclonal antibodies for many viruses causing diseases provides a new momentum in this emerging technology. I will describe fundamental approaches for the development of vaccines using genetically engineered Inoviruses.