Day 2 :
Keynote Forum
Lallan Giri
Biologic Resources LLC, USA
Keynote: Conjugate rPA-PGA anthrax vaccine induces protective antibodies
Time : 10:00-10:40

Biography:
Lallan Giri is currently the CEO of Biologics Resources LLC (BRLLC) which is a Vaccine and Biopharma Company focused on the development of biodefense vaccines for adults and children. He is a Vaccinologist and has made contributions to the development of several pediatric vaccines as Director at Glaxo Wellcome, Director at Sanofi Pasteur, Vice President at Emergent Bio-solutions, and CEO at BRLLC. The current ongoing research projects at BRLLC include work on “Biodefense vaccines and therapeutics”.
Abstract:
Anthrax is a lethal infectious disease caused by Bacillus anthracis. The two major virulence factors of B. anthracis are the poly-γ-D-glutamic acid (PGA) capsule and the exotoxin. Three components of the exotoxin form two bipartite toxins causing edema and organ failure. The anti-phagocytic PGA capsule disguises bacilli from macrophages, allowing unimpeded growth in the host. PA is a target of anthrax vaccine development as it elicits a toxin-specific protective immune response; however PA based anthrax vaccines only address toxin-induced disease and not capsule mediated virulence. Recent effort has been made to include PGA in 2nd generation anthrax vaccines, resulting in a promising vaccine providing comprehensive protection. Antibody to PGA fully neutralizes the capsule but PGA must be conjugated to a carrier protein to provide immunogenicity against both antigens. We have undertaken advanced development of an rPA-PGA conjugate vaccine for adult and pediatric populations. Our hypothesis is that by targeting the two virulence factors of B. anthracis, we can create an anthrax countermeasure that is more comprehensive and efficacious than currently available vaccines. We have shown by ELISA that our conjugate vaccine candidate produces significant levels of antibody to PA and PGA in mouse and rabbit immunogenicity studies. Compared to the approved anthrax vaccine, antibodies produced in response to the rPA-PGA vaccine are more protective in the toxin neutralization assay. We have produced cGMP vaccine and performed a challenge study in rabbits with this vaccine lot, demonstrating 100% protection against lethal aerosol B. anthracis spore challenge in vaccinated rabbits. There is also an increasing need to develop an anthrax vaccine for use in children. We have developed a neonatal rabbit model to simulate the pediatric immune response, showing three week old rabbits generate antibody to both antigens in response to immunization with rPA-PGA that is neutralizing in TNA.
Keynote Forum
Leondios G Kostrikis
University of Cyprus, Cyprus
Keynote: Development of vaccines using genetically engineered Inoviruses
Time : 10:40-11:20

Biography:
Leondios G Kostrikis is a Professor at University of Cyprus. He completed his BSc and PhD at New York University and his Post-doctoral training at Aaron Diamond AIDS Research Center of Rockefeller University. He joined the Rockefeller University as an Assistant Professor in 1999 and the University of Cyprus as Professor in 2003. He was a Fulbright, Elizabeth Glazer Pediatric AIDS Foundation and Aaron Diamond Foundation Scholar. He has directed over 20 competitive grants from the NIH and the European Commission. He is an Editorial Board Member of 12 international journals.
Abstract:
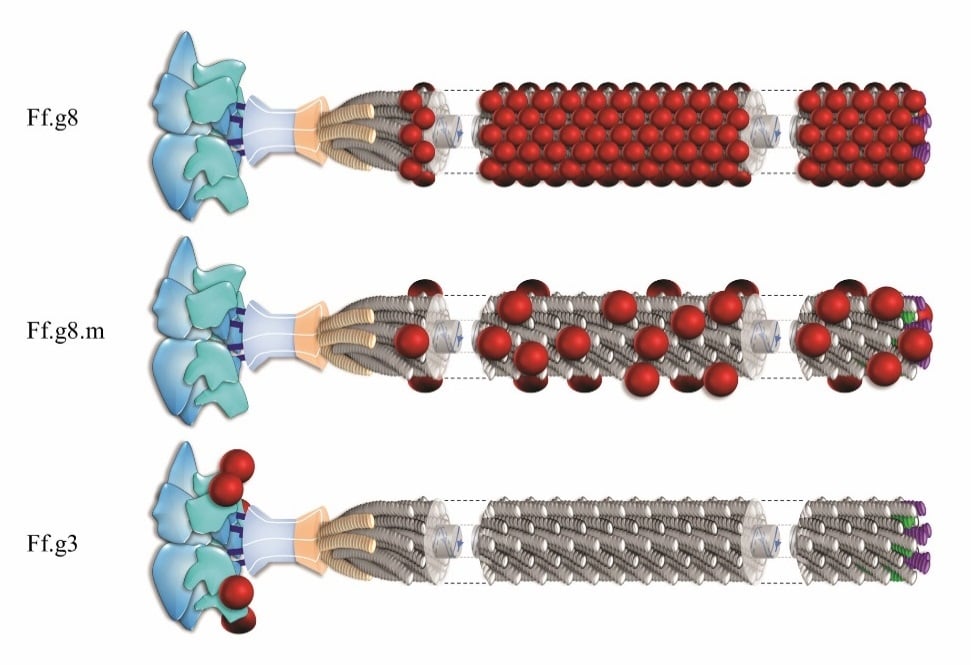
An effective vaccine will likely depend on success in designing immunogens that elicit potent neutralizing antibodies to naturally circulating strains of infectious agents including viruses. In the past, antigen-presenting viral vectors have been extensively used as vehicles for the presentation of antigens to the immune system in numerous vaccine strategies. In previously known vaccine development efforts, two main viral vectors have been used as antigen carriers: Live attenuated vectors and virus-like particles (VLPs); the former, although tends to be highly effective in animal studies, raises safety concerns and the latter is less effective to induce neutralizing antibodies. For more than two decades, Inoviruses have also been utilized as antigen carriers in several vaccine studies. Inoviral vectors are important antigen-carriers in vaccine development due to their ability to present an antigen on their outer architecture in many copies and to their natural high immunogenicity. Numerous fundamental studies have been conducted, which have established the unique properties of antigen-displayed inoviral vectors. The recent isolation of new, potent neutralizing monoclonal antibodies for many viruses causing diseases provides a new momentum in this emerging technology. I will describe fundamental approaches for the development of vaccines using genetically engineered Inoviruses.
- Biodefense Vaccine against Bioterrorism | Vaccines for Immune Mediated Diseases | Vaccines for Pregnant Women and Neonates | Antibodies : Engineering and Therapeutics | Animal Models and Clinical Trials
Location: Sylt 1-2

Chair
Leondios G Kostrikis
University of Cyprus, Cyprus

Co-Chair
Lallan Giri
Biologic Resources LLC, USA
Session Introduction
Lallan Giri
Biologics Resources LLC, USA
Title: Protection against bioterrorism
Time : 11:35-12:00

Biography:
Lallan Giri is currently CEO of Biologics Resources LLC (BRLLC) which is a Vaccine and Biopharma Company focused on the development of biodefense vaccines for adults and children. Professionally, he is a Vaccinologist and has made contributions to the development of several pediatric vaccines during his employment as Director of Glaxo Wellcome, as Director at Sanofi Pateur, and as Vice President at Emergent Biosolutions, Inc, and as CEO at BRLLC.
Abstract:
Bioterrorism involves the intentional release of dissemination of deadly biological agents in the cities and communities. Terrorism has become a common and easier way of causing harms to innocent public. At present time, it has caused serious concerns and insecurity in the public domain. One of the sources or methods used by terrorists is through dissemination of deadly bacteria, viruses, and chemicals. Bioterrorism agents are found typically in nature and can be grown easily. It is also possible that they can be mutated or altered to increase their ability to cause deadly diseases causing mass casualty of human lives. Anthrax letter attacks after September 11, 2001 demonstrated the ease of using the anthrax spores as a weapon of mass destruction in a bioterrorist attack. Anthrax is lethal infectious disease caused by the spore forming Bacillus anthracis. The two major virulence factors of B. anthracis are the poly-y-glutamic acid (Y-D-PGA) capsule and exotoxin. The three components of the exotoxin, Protective Antigen (PA), Lethal Factor (LF) and Edema Factor (EF) are produced and secreted separately and form two bipartite toxins that cause massive edema and organ failure. The anti-phagocytic y-D-PGA capsule disguises the bacilli from immune surveillance by macrophages and allows unimpeded growth of the bacilli leading to anthrax disease caused by both, the capsule and toxins. However, the currently licensed Anthrax Vaccine Adsorbed (AVA) or rPA based anthrax vaccines only address the toxin- induced disease, and not the capsule mediated virulence factors related disorders which contribute to the severity of the anthrax infection leading to death. The newly proposed anthrax vaccine, an antibody response triggered specially by y-D-PGA can fully neutralize the disguising capsule. However, the capsule is not immunogenic by itself and must be conjugated to a carrier protein such as PA. The conjugate vaccine concept embodies the paradigm of combining both, antibacterial (prophylactic) and antitoxin (therapeutic) components into a single vaccine. This has contributed much success in inducing protective levels of antibodies in infants and children against systemic infection with encapsulated pathogens. The development of this conjugated anthrax vaccine will fill an urgent void, delivering a well-defined and characterized biodefense countermeasure potentially suitable for infants and children. The proposed work will lead to the characterization, testing, and manufacturing of a promising conjugated anthrax vaccine candidate. This will potentially lead to the development of a conjugated anthrax vaccine with dual antigens, namely PA and PGA (polyglutamic acid).
Mark Lawrence Johnson
MJ Lawrence Consulting, Germany
Title: When funding for medical countermeasures against rare, but detrimental agents is cost-effective
Time : 12:00-12:25

Biography:
Mark Lawrence Johnson is one of the early pioneers to develop policy and procurement in the European biodefense market for medical countermeasures (MedCM). He has accrued a mature and extensive network. His professional credibility was born over 20 years ago when he began to represent leading global pharmaceutical companies such as Merck & Co., Eli Lilly, and Astellas. Following response to the European Commission’s (EC) Green Paper on CBRN preparedness in 2007, his participation at EC CBRN taskforce meetings was instrumental in getting fundamental economic elements adopted in its corresponding recommendations and action plan. He holds an MBA from Wake Forest University and a BA in Business Management from the University of Maryland. He is currently a Doctoral candidate at the Paris 2 University, Panthéon-Assas (LEMMA) working to determine economic mechanisms for achieving international availability of CBRN MedCM.
Abstract:
Some chemical, biological, radiological, and nuclear (CBRN) agents can be very damaging and pose high risk to national security because they bear potential to induce economic and social disruption. Assuming efficacious medical research and development (R&D) is executed; populations could be protected against such CBRN agents via new prophylactic drugs and vaccines or post-exposure treatment with antidotes and antimicrobials. However, the development of medical countermeasures (MedCM) against these agents is very limited. This presentation depicts specific features of the MedCM market and R&D against CBRN agents and explores the latest Ebola outbreak as an illustration of these concerns. While their R&D process and related costs are quite similar to those of commercially viable conventional diseases, it is shown that free market rewards and incentives are more uncertain and most often not sufficient for developers of MedCM to independently invest on its own since widespread dissemination of CBRN agents remains a rare and unpredictable event (e.g. few customers, low and volatile market sales potential). This results in market failures for appropriate medicines or vaccines except in the case of effective public intervention. However, priorities should be identified given the extent of such risks and the amounts of investment involved. The Ebola example is used as a case study of causal factors which led to a lack of a vaccine prior to the 2014 outbreak. In addition, possible reasons which may have triggered a re-evaluation of its prioritization as a threat worthy of high actionable concern are probed.

Susanne Rauch
CureVac, Germany
Title: An mRNA-based vaccine technology for next generation prophylactic vaccines
Time : 12:25-12:50

Biography:
Susanne Rauch works as a Scientist at CureVac where she is involved in the development of mRNA-based prophylactic vaccines. She has been trained as a Postdoc at King’s College London and as a PhD student at University Hospital Heidelberg where her work was focused on “The biology of different viruses such as HIV-1, MLV and herpesviruses.
Abstract:
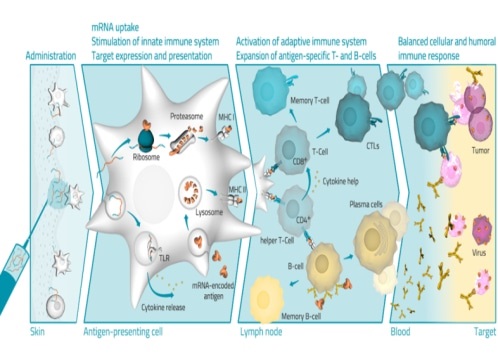
In recent years, messenger RNA (mRNA) based technologies have increasingly been applied in vaccine development. Such approaches have utilized mRNA for both therapeutic cancer vaccinations, and for prophylactic vaccines, drawing much attention from industrial and academic fields. RNActive®, an mRNA based vaccination technology, has yielded promising results in the development of vaccines against a variety of viral pathogens such as RSV (respiratory syncytial virus), influenza, rabies, and Ebola virus in several animal models. We have previously shown that intradermal (i.d.) application of RNActive® vaccines is able to confer protection against lethal influenza and rabies virus challenge infection in mice and induces protective levels of functional antibody responses against both viruses in domestic pigs. However, having been optimized for i.d. application, immunological responses upon intramuscular (i.m.) injection of these mRNA vaccine formulations remained less efficient as yet. Here, we describe an alternative formulation of RNActive® vaccines that is able to induce potent immune responses when applied intramuscularly using low doses (µg) of mRNA. Vaccination of mice with this RNActive® formulation encoding for influenza HA (hemagglutinin) or rabies G (glycoprotein) revealed an increase of both humoral and cellular immune responses, analyzed via functional antibody levels and ICS (intracellular cytokine staining) of T cells, respectively, compared to previous RNActive® formulations. Further experiments showed that this new vaccine formulation was able to induce potent and long lasting immune responses against influenza HA as well as high titers of rabies virus neutralizing antibodies in NHPs (non-human primates).
Shreemanta K Parida
Justus-Liebig University, Germany
Title: ATMP Cell Therapy: Triumphs and constraints for unmet clinical needs
Time : 12:50-13:15

Biography:
Shreemanta Parida is a Clinician Scientist with his expertise and passion in making a difference to patients by applying cross cutting scientific advances in patient care. He has been engaged during last 5 years in bringing ATMP cell therapy to translation in precision medicine for many unmet clinical needs. He has contributed immensely to the field of Global Health, Vaccines and Immunology over last three decades with focus on resource poor settings embracing stakeholders across disciplines. He has held many leadership positions in many top institutions in Europe, Africa and India.
Abstract:
Immunotherapy for Cancer, adjudged "Breakthrough of the year 2013" by journal "Science", was a paradigm shift and "game changer" in the conquest of cancer by targeting immune-system than the tumour. Cell therapy and immune therapy have opened up plethora of options to address unmet clinical needs with greater role in haemato-oncology field with promising clinical success in combating graft vs. host disease in transplant patients as well as in preventing infections in them. However, achieving the optimal numbers and the desired phenotypes of cells by conventional expansion culture methods have been major constraints. The increasing use of cell therapy towards myriads of clinical conditions has led to production processes in accordance with Good Manufacturing Practice (GMP) for Advanced Therapeutic Medicinal Product (ATMP). In cellular therapy, safety remains of paramount importance and refers to consistency, quality and potency, not only at the batch release level but also during the process development which should be adapted to closed systems that are easy to use. Implementing dynamic controls during the manufacturing of clinical-grade cells for therapy is essential to ensure microbiological safety and to avoid potential adverse effects linked to genomic instability driving transformation and senescence or decrease of cell functions (immunoregulation, differentiation potential). To meet this growing need of producing required cells of choice in bulk from bone marrow, peripheral blood or other tissues consistently in quality and numbers in a controlled, reproducible, robust, and efficient dynamic environment, tools and technologies have evolved offering in the form of GMP certified closed system at affordable cost to advance cell therapy into practice for the unmet clinical needs. We are at an interesting crossroad to push the limits making it accessible for the best outcomes for patients in precision medicine by validating tools and technologies as well as performing robustly designed clinical trials as way forward.

An Overview of Manufacturing possibilities for ATMP Cell Therapy
Guanggang Qu
Shandong Binzhou Animal Science and Veterinary Medicine Academy, China
Title: Efficient expression of porcine circovirus type 2 virus-like particles in Escherichia coli
Time : 14:30-14:55

Biography:
Guanggang Qu is interested in Preventive Veterinary Medicine, Veterinary Biotechnology and Biological Products. He is particularly interested in developing innovated vaccines by E. coli expression system. Currently, he is an Assistant Professor at Shandong Binzhou Animal Science & Veterinary Medicine Academy and Vice Director at Shandong Binzhou Research and Development Centre of propolis vaccine for livestock and poultry. He was a Visiting Scientist at Bhabha Atomic Research Centre and at U.S. Department of Agriculture of USA from 2011 to 2013. He completed his Doctoral Degree at Jilin University, China.
Abstract:
Porcine circovirus 2 (PCV2) is considered to be the etiologic agent responsible for porcine circovirus associated disease (PCVAD) that frequently affects growing pigs at 5-18 weeks of age and can lead to significant negative impacts on profitability of pork production. The capsid (Cap) protein of PCV2 is a major candidate antigen for development of recombinant vaccine and has been successfully used as a vaccine to control porcine circovirus associated disease (PCVAD). In our study, full-length ORF2 gene with codon-optimized for E. coli was synthesized and inserted into the PTF9 (+) expression vector to improve expression of recombinant Cap protein (rCap). A large amount of soluble rCap protein was obtained. The recombinant Cap protein expressed by Escherichia coli (E. coli) has the ability to self-assemble into virus-like particles (VLPs) in vitro; it is particularly an opportunity to develop the PCV2 VLPs vaccine in E coli. In this report, a highly soluble Cap-tag protein expressed in E coli was constructed with a p-TF9 expression vector with a fusion tag of TF9. The recombinant Cap was purified using Ni2+ affinity resins and the tag was removed by the TEV protease. Simultaneously, the whole native Cap protein was able to self-assemble into VLPs in vitro when viewed under an electron microscope. The Cap-like particles had a size and shape that resembled the authentic Cap. The result could also be applied in the large-scale production of VLPs of PCV2 and could be used as a diagnostic antigen or a potential VLP vaccine against PCV2 infection in pigs. We have, for the first time, utilized the Fh8 fusion motif to successfully express the complete Cap protein of PCV2 in E coli. After the cleavage of the fusion motif, the nCap protein self-assembled into VLPs, which can be used as a potential vaccine to protect pigs from PCV2-infection.
Joseph L Mathew
Postgraduate Institute of Medical Education and Research, India
Title: Comparison of susceptibility to measles in preterm infants versus term infants
Time : 14:55-15:20

Biography:
Statement of the Problem: In India and many other developing countries, a single dose of measles vaccine is administered to infants at 9 months of age. This is based on the assumption that maternal transplacentally transferred antibodies will protect infants until that age. However, our previous data showed that most infants lose maternal anti-measles antibodies before 6 months of age, making them susceptible to measles before vaccination at 9 months. This prospective study was designed to compare susceptibility in pre-term vs. term infants, at different time points.
Methodology: Following Institutional Ethics Committee approval and a formal informed consent process, venous blood was drawn from a cohort of 45 consecutive term infants and 45 consecutive pre-term infants (both groups delivered by the vaginal route); at birth, 3 months, 6 months and 9 months (prior to measles vaccination). Serum was separated and anti-measles IgG antibody levels were measured by quantitative ELISA kits (with sensitivity and specificity >95%). Susceptibility to measles was defined as antibody titre <200 mIU/ml. The mean antibody levels were compared between the two groups at the four time points.
Findings: The mean gestation of term babies was 38.5±1.2 weeks; and pre-term babies 34.7±2.8 weeks. The respective mean birth weights were 2655±215 g and 1985±175 g. Reliable maternal vaccination record was available in only 7 of the 90 mothers. Mean antimeasles IgG antibody (±SD) in terms babies was 3165±533 IU/ml at birth, 1074±272 IU/ml at 3 months, 314±153 IU/ml at 6 months and 68±21 IU/ml at 9 months. The corresponding levels in pre-term babies were 2875±612 IU/ml, 948±377 IU/ml, 265±98 IU/ml and 72±33 IU/ml at 9 months (p>0.05 for all inter-group comparisons). The proportion of susceptible term infants at birth, 3 months, 6 months and 9 months was 0%, 16%, 67% and 96%. The corresponding proportions in the pre-term infants were 0%, 29%, 82% and 100% (p>0.05 for all intergroup comparisons).
Conclusion & Significance: Majority of infants are susceptible to measles before 9 months of age suggesting need to anticipate measles vaccination, but there was no statistically significant difference between the proportion of susceptible term and pre-term infants, at any of the four time points. A larger study is required to confirm these findings and compare sero-protection if vaccination is anticipated to be administered between 6 and 9 months.
Abstract:
Joseph L Mathew works at the Advanced Pediatrics Centre, Postgraduate Institute of Medical Education and Research, Chandigarh, India. He has contributed extensively to evidence-based policy-making for several vaccines in the Indian context, especially Hepatitis B, Hib, IPV, MMR, PCV, Influenza, Varicella, acellular pertussis, HPV, Rotavirus, and typhoid conjugate vaccines. He is one of the first to identify the rapid waning of maternal measles antibodies in infancy, creating a pool of susceptible infants/children. He has nearly 200 peer-reviewed publications to his credit and delivered numerous presentations related to vaccinology in national and international meetings.
Aamir Shaikh
Assansa, India
Title: Vaccines for key unmet Medical (Infectious Disease) needs in the developing world: Learning and reflections from the India Polio eradication success story
Time : 15:20-15:45

Biography:
Aamir Shaikh is a “Pharmaceutical Physician” who began his health care industry career in 1998 as a Medical Advisor, with Pfizer India. His early experience with vaccines was related to educational and promotional endeavors with a Hepatitis B vaccine in India. Over the next decade, he successfully delivered in positions of increasing responsibility and leadership, culminating in his role as Director, Medical Affairs & Research for Pfizer India. In 2007, he moved on from Pfizer to start his own small Health Care Consultancy, Assansa. Throughout his health care career, and now through Assansa, his key endeavor is “Helping Health Care Professionals Grow.” He believes this can be achieved through an identified personal philosophy and mindset - one which embraces knowledge, science, business, and ethics.
Abstract:
Over the 20th century, vaccination has been the most effective medical intervention to reduce morbidity and mortality caused by infectious diseases. It is estimated that vaccines save around 2–3 million lives per year worldwide. However, even today, we do not have vaccines to prevent many noteworthy infectious diseases, and access issues preclude available vaccines from being utilized to their fullest potential. Key unmet medical (infectious disease) needs, more so for the developing world, include HIV, malaria, TB, dengue, and others. The Association of Southeast Asian Nations (ASEAN) is responding to these unmet needs through the efforts of the ASEAN-Network for Drugs, Diagnostics, Vaccines, and Traditional Medicines Innovation (ASEAN-NDI). This is important considering that infectious tropical diseases remain prevalent, emerging, and reemerging in the region. India has invested significant health resources towards its ambitious Polio Eradication Program, and is now enjoying the fruits of success. Effectively using the Oral Polio Vaccine (OPV), the key pillars of the Polio Eradication strategy, have visibly demonstrated effective results, and have provided us with useful learning. Moreover, the innovative creation and use of Social Mobilization Network (SMNet) has also provided us with important conceptual and practical lessons for health communication, social mobilization, and for partnerships in global health. Over 2017, and beyond, India will have to grapple with significant challenges - these will need to be addressed with a sense of vigilance and perseverance. There is reason to hope that the challenges of infectious diseases in the 21st century will be adequately addressed by the research, development, access and use of vaccines. Beyond the science, success will largely depend on the “human connect”, and will be facilitated through shared vision, common purpose, effective partnerships, and sound leadership.
Liliya Pekova
University hospital (Stara Zagora), Bulgaria
Title: Pneumococcal meningoencefalitis in 50-day-old baby ended with lethal outcome: A case report
Time : 15:45-16:10

Biography:
Liliya Pekova is a Head of Clinic of Infectious Diseases at University Hospital in Stara Zagora, Bulgaria. She is a Chief of Department of Infectious Diseases, Medical Faculty at Trakia University. Her scientific interests include “Viral hepatitis, neuro-infections and tick-borne diseases”. The main topic of her researches is Acute Hepatitis C. She has two specialties: Infectious diseases and Epidemiology. She is interested in area of Vaccines.
Abstract:
Streptococcus pneumoniae (S. pneumoniae) is a usual agent of pneumonia, sinusitis and medial otitis. In children under two-year-old, it appears as a second bacterial agent which is responsible for neuro infections. We presented a case of S. pneumoniae meningoencephalitis as a primary localization in a 50-day-old breast-fed child. The illness began with high temperature and mild catarrh. Three days later convulsions without meningeal signs were demonstrated. Diagnosis was based on cerebral fluid changes and microbiological verification. In spite of triple antibacterial starting suitable to microbial sensitivity, neuro infection completed with lethal outcome. Laboratorial data–low levels of natrium, severe acidosis in conjunction with high level of protein, cells and low glucosis were considered as prognostic signs for unfavorable outcome. The baby was not vaccinated with synflorix because his age was small. Possibilities to use vaccine against S. pneumoniae earlier than usual were discussed.
Marine Meunier
French Agency for Food, Environmental and Occupational Health & Safety, France
Title: Promising results in the assessment of new vaccine candidates against campylobacter in poultry
Time : 16:10-16:35

Biography:
Marine Meunier completed her Master’s in Immunology and Vaccinology and 7-month internship at GSK Vaccine, and PhD at French Agency for Food, Environmental and Occupational Health & Safety (ANSES) in the Laboratory of Ploufragan. The thesis project in which she is involved is a part of European CAMPYBRO project about control of Campylobacter infection in broiler flocks through two-steps strategy: Nutrition and vaccination. She developed the tools to assess the vaccine potential of these antigens (development of an avian vaccine protocol against Campylobacter and ELISA tests). She used molecular biology tools and FLPC for the production and purification of vaccine antigens.
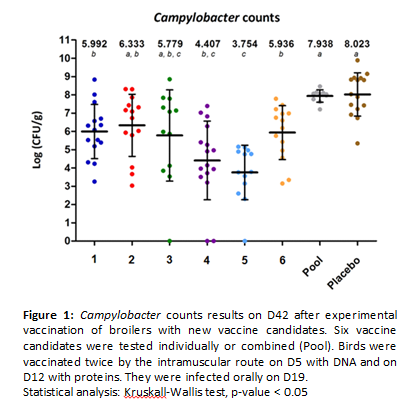
Abstract:
Campylobacteriosis is the most prevalent human bacterial gastrointestinal disease in Europe. Birds are the main reservoir of Campylobacter and human contaminations principally occur by consumption and handling of poultry meat. It was estimated that a Campylobacter reduction from 2 to 3 log10 CFU/g of the intestinal contents in live broilers could be responsible for a decrease of 76% to 100% of the infection in humans. Vaccination of poultry could be a potential way in this goal but despite many studies, no efficient vaccine is available yet and research of more powerful vaccine antigens against Campylobacter is needed. The recent in silico analysis of Campylobacter genome using the reverse volcanology strategy allowed the identification of 14 potential new vaccine candidates. After developing an avian vaccine protocol consisting in two immunizations by the intramuscular route on day five with DNA and day 12 with proteins followed by an oral challenge on day 19, we assessed in vivo immune and protective powers of six new vaccine candidates, individually or in combination. Among the six antigens, four had a significant effect on both IgY production in serum and reduction of Campylobacter caecal counts. The mean reduction of caecal counts with those four proteins varied from 2 to 4.2 log10 CFU/g of caecal content. The pooled proteins had no protective effect. A new in vivo experiment is actually on going in order to confirm these results. The first results are promising since they reach reductions better than those estimated to impact human diseases incidence. Also these results proved the interest of the reverse vaccinology approach to screen new candidate proteins that may become efficient vaccines.